HC-5404
HC-5404
HC-5404
Clinical Development of HC-5404
----------
Inhibition of PERK by HC-5404 results in significant antitumor efficacy as combination with varied standard of care anti-angiogenic agents in multiple solid tumor models, including renal cell carcinoma and gastric cancer.
HC-5404 recently completed investigation in a Phase 1a trial as a monotherapy for the treatment of advanced solid tumors enriched for patients with renal cell carcinoma (RCC) and gastric cancer. Following the completion of our Phase 1a trial, we are now seeking partners for a Phase 1b trial in combination with VEGFR TKIs in multiple solid tumors.
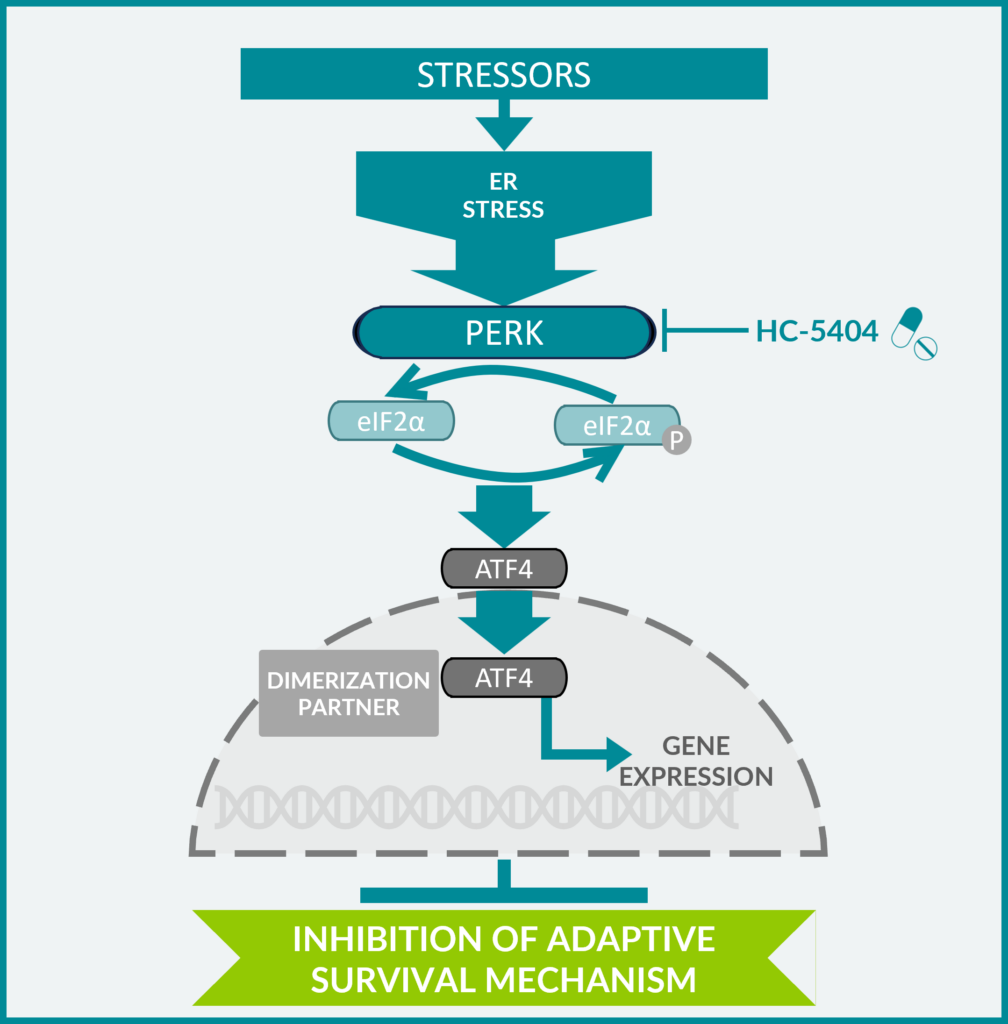
PERK and the Unfolded Protein Response
- The Unfolded Protein Response (UPR) is an adaptive cellular program used by cancer cells to survive by facilitating:
- Adaptation to harsh tumor microenvironment characterized by hypoxia, nutrient deprivation, oxidative stress
- Hypoxia drives accumulation of misfolded proteins in the endoplasmic reticulum (ER), leading to ER stress and activation of PERK (protein kinase R–like endoplasmic reticulum kinase) in the UPR pathway
- PERK is an ER-resident transmembrane kinase that signals through eIF2a to block ER-dependent protein synthesis and maintain homeostasis
- Various SOC cancer therapies (i.e. VEGFR TKIs) further amplify ‘stress’ in tumors and activate PERK as a survival mechanism
- HC-5404, a First-in-Human PERK inhibitor, has completed a Phase I clinical trial focused on solid tumors
- Inhibition of the adaptive UPR via PERK inhibition results in monotherapy and combinatorial anti-tumor activity with anti-angiogenics in multiple tumor types